Phylogenetic and genetic evidence suggests that the complex plays a broader role beyond stimulation of the phosphorelay.īacillus subtilis is a Gram-positive bacterium that can form spores, develop biofilms and become competent to take up extracellular DNA from the environment. 
This work provides the first comprehensive characterization of an iron-sulfur protein complex that regulates Spo0A∼P levels. As with other iron-sulfur cluster-binding proteins, the complex was also found to bind FAD, hinting that these cofactors may be involved in sensing the cellular redox state. 
In this study, we demonstrate that two 2+ clusters can be assembled on the complex. Indeed, this complex is sufficient to stimulate the rate of phosphotransfer amongst the phosphorelay proteins in vitro. These proteins directly interact and form a stable complex, which has been proposed to accelerate the phosphorelay. RicA, RicF and RicT (previously YmcA, YlbF and YaaT) have been shown to be important regulatory proteins for multiple developmental fates. 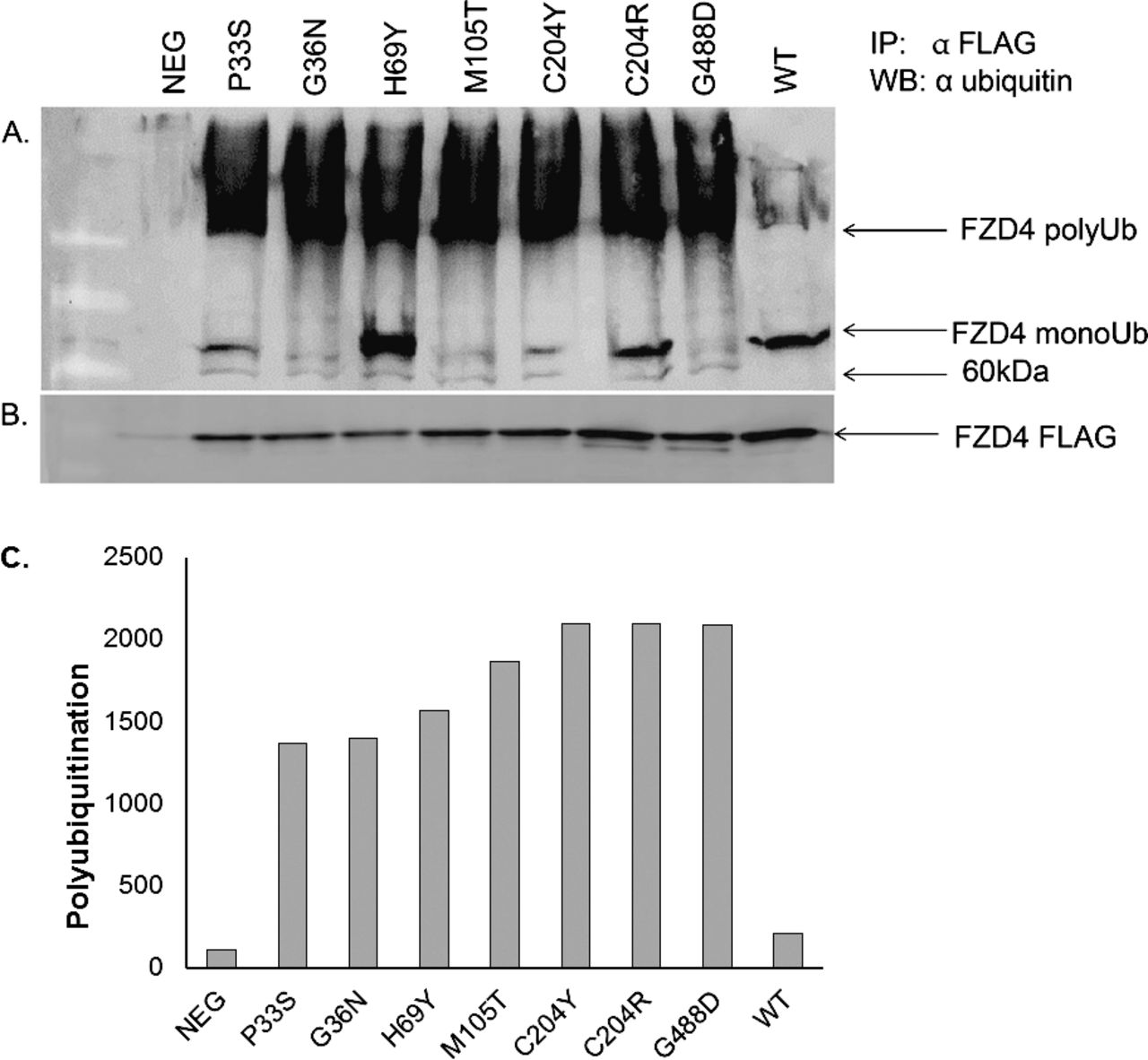
The phosphorylation state of Spo0A is controlled by a multi-component phosphorelay. Each of these states is regulated in part by the phosphorylated form of the master response regulator Spo0A (Spo0A∼P). During times of environmental insult, Bacillus subtilis undergoes developmental changes leading to biofilm formation, sporulation and competence.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
